 Medovia Medical
Medovia Medical
 Medovia Medical
Medovia Medical
Ensuring sterile conditions for surgical tools in China is paramount for patient safety. In the words of Dr. Li Wei, a renowned expert in surgical practices, "Sterility is not just a guideline; it is the foundation of successful surgeries." This highlights the critical nature of maintaining a sterile environment in a surgical setting.
The process of achieving sterility involves strict adherence to established protocols. Factors such as tool cleaning, sterilization techniques, and environmental controls must be meticulously followed. The challenge lies in ensuring compliance across all levels. Many facilities struggle with understanding "How to maintain sterile conditions for surgical tools?" It requires ongoing education for staff and systematic checks.
Moreover, cultural practices and resource limitations may complicate adherence to these standards. Workshops and training can help bridge the gap, yet disparities in understanding still exist. This reflects a need for continuous improvement and accountability. The pursuit of perfect sterility is an ongoing journey, demanding constant vigilance and commitment.
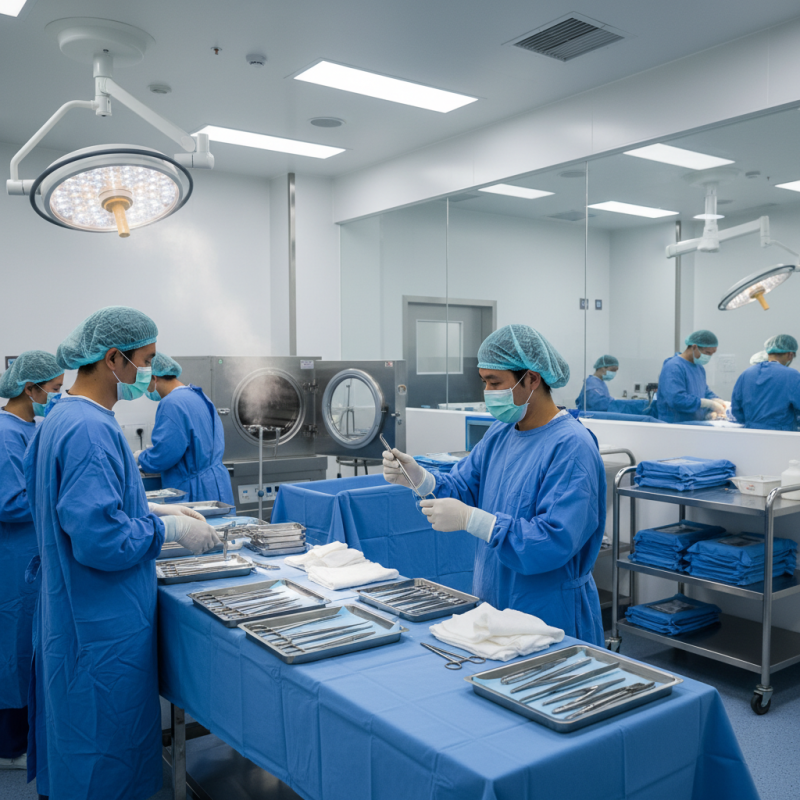
Sterility in surgical procedures is crucial. Contaminated tools can lead to infections, prolong recovery, or even result in complications. Every surgical environment must prioritize sterility to protect patients and ensure successful outcomes. In China, hospitals and clinics face unique challenges in maintaining these conditions due to varying resources and practices.
Achieving sterility involves meticulous cleaning and sterilization protocols. Tools must be properly washed before being autoclaved or subjected to chemical sterilization. Environmental factors such as humidity and air flow must also be controlled. However, inconsistency in practices is a common issue. Some facilities may not adhere strictly to established guidelines. This inconsistency can lead to dangerous outcomes, making it essential for healthcare providers to evaluate their sterilization processes regularly.
Training medical staff is another critical factor. Understanding the importance of sterility ensures that everyone involved in surgical procedures is aware of their role in maintaining safety. Yet, training programs may not always be up to date, which can hinder effective practices. Hospitals must invest in continuous education, emphasizing the significance of sterile tools and environments in patient health.
Ensuring sterile conditions for surgical tools is crucial in any healthcare setting. Key principles of sterilization are foundational for patient safety. Proper cleaning is the first step. Surgical instruments should be scrubbed to remove visible debris. This reduces the likelihood of contamination before sterilization.
Choosing the right sterilization method is vital. Steam sterilization, chemical agents, and dry heat are common techniques. Each has its specific uses and effectiveness. It's essential to follow the manufacturer's guidelines for each tool. However, not all methods guarantee complete sterilization. Regular validation of the sterilization process can reveal gaps. Monitoring sterilization indicators can provide peace of mind.
Training staff on sterilization protocols leads to fewer mistakes. Not all medical personnel may fully understand the complexities involved. Regular workshops and seminars can reinforce best practices. Sterile tools may not always remain sterile during storage or transportation. Improper handling can lead to new contamination risks. Maintaining awareness and vigilance is part of the ongoing process.
| Sterilization Method | Description | Advantages | Limitations |
|---|---|---|---|
| Steam Sterilization | Uses high-pressure steam to eliminate all forms of microbial life. | Effective against a wide range of microorganisms, quick cycle time. | Not suitable for heat-sensitive instruments. |
| Ethylene Oxide (EtO) Sterilization | Uses ethylene oxide gas to sterilize sensitive instruments. | Suitable for heat-sensitive items, effective at low temperatures. | Long cycle times, toxic gas requires careful handling. |
| Radiation Sterilization | Uses gamma rays or electron beams to sterilize products. | Effective for large volumes, no residuals on products. | High initial setup cost, requires specialized facilities. |
| Dry Heat Sterilization | Uses hot air for sterilization, ideal for metal instruments. | Effective for items that can tolerate high temperatures. | Longer time and higher temperatures required than steam. |
Maintaining sterile conditions for surgical tools is crucial in healthcare settings. Cleaning and disinfecting surgical instruments require a systematic approach. Begin by rinsing instruments immediately after use. This helps prevent blood and tissue from drying, making later cleaning easier. Use an enzymatic cleaner to break down organic materials effectively.
Following this, instruments should be thoroughly scrubbed with a soft brush. Pay close attention to hinges, joints, and any crevices where contaminants may hide. Rinsing instruments under running water ensures that all cleaning agents are removed. After cleaning, ultrasonic cleaning can be beneficial for stubborn residues.
Disinfection follows cleaning. Steam sterilization is a reliable method, but it might not reach every surface. Ethylene oxide gas may also be used for delicate instruments that cannot withstand high heat. Regular maintenance and staff training are vital. If cleaning protocols are not properly followed, the risk of infection increases. Reflecting on processes can reveal gaps that need attention, ensuring better outcomes for patients.
Sterile conditions for surgical tools are critical in preventing infections. Validation methods play a crucial role in achieving this. Various techniques can be employed to assess the sterility of surgical instruments.
One effective validation method is physical monitoring. This includes using temperature and pressure indicators during sterilization processes. These indicators help verify that tools have reached the necessary conditions for effective sterilization. However, it is essential to note that relying solely on indicators may not capture every potential failure point. Regular checks and maintenance of sterilization equipment are vital to ensuring reliability.
Chemical indicators are another option. They change color to show whether the tools underwent proper sterilization. Yet, these indicators do not guarantee sterility on their own. They should be part of a broader validation strategy. Additionally, biological indicators, which use live spores, provide the most reliable validation. They confirm that the sterilization process was successful. Nonetheless, they require careful handling and interpretation. The process is not infallible, and mistakes can occur, emphasizing the need for continuous improvement in validation methods.
Sterilization in China's healthcare system is critical for ensuring patient safety. The regulatory standards are stringent and aim to minimize infection risks. Various methods are utilized for sterilization, including steam sterilization and ethylene oxide. Hospitals must follow these guidelines closely. However, compliance is inconsistent. Some facilities struggle with maintaining updated technologies and proper training among staff.
The China Food and Drug Administration oversees sterilization regulations. They provide protocols that healthcare providers must follow. Regular audits are conducted to ensure adherence, but gaps still exist. Many hospitals report challenges in keeping up with evolving standards. Resources may be limited, impacting the effectiveness of sterilization programs.
Emphasizing education is essential. Continuous training ensures that staff are familiar with the latest practices. Additionally, investing in advanced sterilization equipment can help meet regulatory demands. Still, the commitment to improving processes is ongoing. Healthcare providers must remain vigilant and responsive to changes in regulations. This responsibility is vital for maintaining sterile conditions and ensuring patient safety.
: Sterile conditions prevent infections and ensure patient safety during surgeries.
Validation methods include physical monitoring, chemical indicators, and biological indicators.
They use temperature and pressure indicators during sterilization to verify proper conditions.
No, chemical indicators alone do not guarantee sterility and should be part of a broader strategy.
Biological indicators using live spores provide the most dependable validation of sterilization.
Compliance can be inconsistent due to outdated technologies and insufficient staff training.
The China Food and Drug Administration is responsible for monitoring sterilization compliance.
Continuous staff education and investment in modern equipment can enhance sterilization effectiveness.
Regular audits are conducted, but many facilities face challenges in maintaining compliance.
Many hospitals struggle with resource limitations that affect their sterilization programs.
Ensuring sterile conditions for surgical tools is critical for the safety and success of surgical procedures. To maintain sterile conditions for surgical tools, it is essential to understand the significance of sterility and adhere to key principles of sterilization. Effective cleaning and disinfecting methods are fundamental practices that help eliminate contaminants from surgical instruments.
Additionally, the validation of sterilization processes is vital to confirm that sterility is achieved consistently. Regulatory standards governing sterilization in China’s healthcare system provide a framework for maintaining these important practices. By following these guidelines, healthcare professionals can ensure that surgical tools are safe for use, thereby protecting patient health and enhancing surgical outcomes.
