Product Overview
Pulsating vacuum sterilizer is strictly designed and manufactured in accord with GMP technical Standard. It has passed ISO9001 quality management qualification standard and ISO13485:2003 Certificate of Quality Management System for Medical Devices. It adopts PLC and man-computer interface automatic program control. The relevant data and curve can be saved and printed automatically.
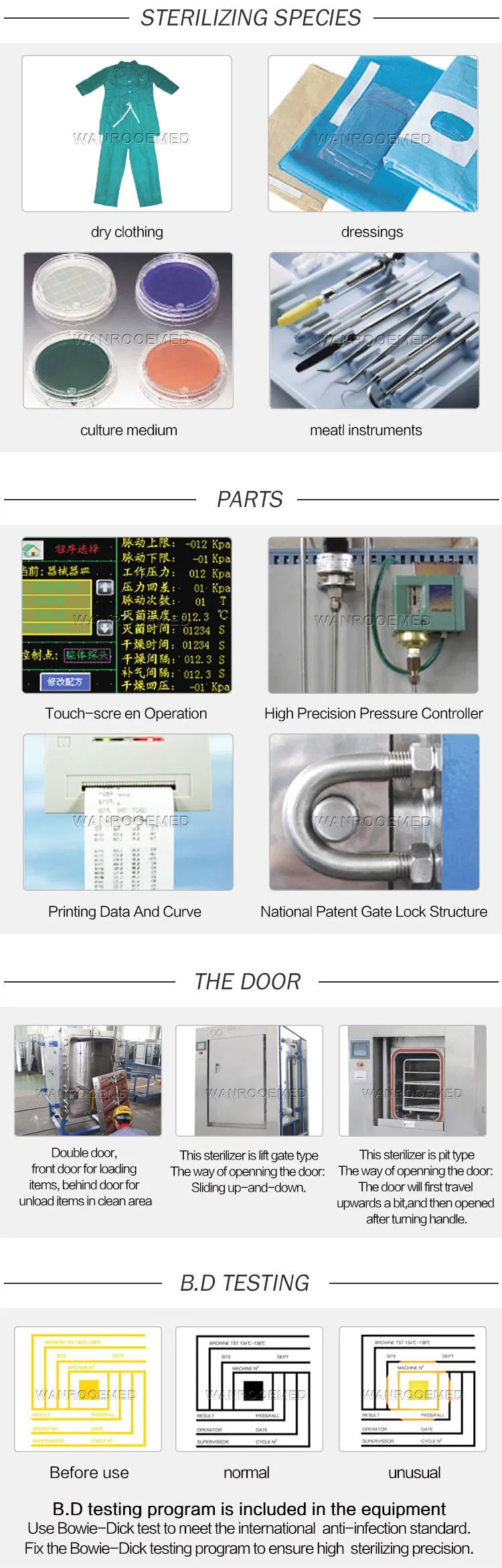
Applications: This autoclave is suitable for pharmaceutical factories, hospitals, scientific research institutes and other units to sterilize and dry asepsis clothes, rubber goods, metal instruments, culture medium, etc.
Sterilization Process Flow
The autoclave supports both automatic and manual operations with preset editable programs for various materials:
Dynamic Pre-vacuum: Removes air with no heat build-up.
Pressure Pulsing: Rapid removal of non-condensable gases and pre-heating.
Sterilization: Steam injection until working pressure and temperature are achieved.
Drying: High vacuum and jacket heat radiation for effective drying.
Cooling: Controlled vacuum break to accelerate the cooling process.
Frequently Asked Questions
Q1: What materials are used for the sterilization chamber?
The sterilization chamber is constructed from high-grade SUS316 stainless steel, while the outer shell is made of SUS304 for durability and hygiene.
Q2: Which control system is integrated into the YG series?
The system utilizes a Siemens PLC and a user-friendly HMI color touch screen for precise automatic program control and data monitoring.
Q3: What items can be sterilized using this pulse vacuum autoclave?
It is ideal for sterilizing surgical instruments, medical cloths, rubber goods, culture media, and other items required in hospitals and labs.
Q4: Does the unit include a drying function?
Yes, the unit features a dedicated drying phase using high vacuum and heat radiation from the jacket walls to ensure materials are thoroughly dried.
Q5: Are there safety mechanisms to prevent improper operation?
Yes, it includes mechanical, electrical, and software interlocks, safety valves on the chamber and jacket, and an emergency E-Stop mechanism.
Q6: Can sterilization data be recorded?
Absolutely. The sterilization data and curves can be printed via a micro-printer or transferred to a USB disk for record-keeping and GMP compliance.





 Medovia Medical
Medovia Medical