 Medovia Medical
Medovia Medical
 Medovia Medical
Medovia Medical
In the modern healthcare ecosystem, the patient bed is no longer just a piece of furniture; it is a sophisticated clinical interface that integrates ergonomics, patient safety, and digital monitoring. As global healthcare demands surge due to aging populations and infrastructure modernization, China has emerged as the world's primary hub for patient bed factories and exporters. Companies like Jiangsu Medovia Medical Co., Ltd. are at the forefront, bridging the gap between high-end clinical requirements and cost-effective manufacturing.
The transition from manual crank beds to fully automated AI-driven ICU platforms represents a significant technical leap. For procurement officers and hospital administrators, choosing the right manufacturer involves evaluating not just the price, but the "Information Gain" regarding life-cycle costs, sterilization compatibility, and long-term mechanical reliability.
Jiangsu Medovia Medical Co., Ltd. is a professional and reliable supplier in the medical equipment industry in China, committed to delivering advanced, high-quality, and cost-effective healthcare solutions to customers worldwide. With years of industry experience, we have built a strong reputation as a trusted partner for medical institutions and distributors.
Our mission is to provide reliable, innovative, and accessible medical equipment manufactured in China. We strive to enhance global healthcare standards through quality products and build long-term partnerships based on trust and mutual growth.
Focus on heavy-duty steel frames, manual height adjustments, and high-load casters. Essential for basic clinical environments and rural healthcare.
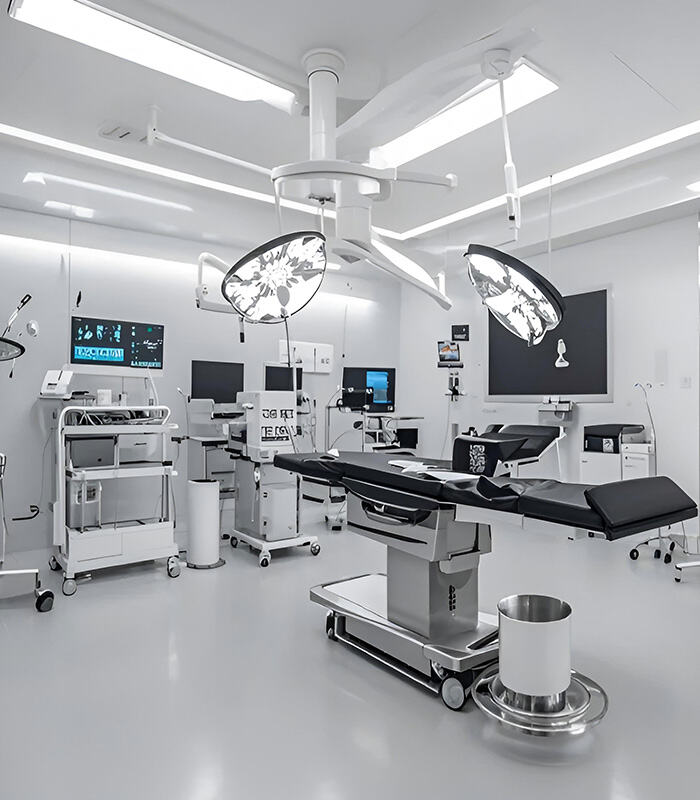
Integration of silent LINAK or T-Motion actuators for Trendelenburg positioning, backrest, and knee-break controls. Enhanced patient autonomy.
Weight sensors for fall prevention, integration with Nurse Call systems, and pressure-redistribution surfaces to prevent bedsores.
| Feature | Standard Ward Beds | ICU Electric Beds | Home Care Solutions |
|---|---|---|---|
| Material | Cold-rolled Steel / ABS | Anti-microbial Nano Coating | Wood-grain Aesthetic / Steel |
| Control System | Manual / Semi-Electric | Fully Integrated Digital Touch | Wireless Remote Control |
| Safety Standard | EN 60601-2-52 | IEC 60601 Medical Grade | Domestic Use Certifications |
Why source from China? The answer lies in the Vertical Integration of the supply chain. From raw steel processing to final electronics assembly, our Jiangsu-based facilities optimize every stage to reduce lead times and ensure consistency.

Automatic Slitting

CNC Lathe

Laser Engraving

Grinding

Ultrasonic Clearing
Quality is at the core of everything we do. All our products are manufactured under strict quality control systems and comply with international standards. We are proud that our products are certified by ISO and CE systems, ensuring safety, stability, and performance in clinical environments. From raw material selection to final inspection, every step of production is carefully monitored to guarantee consistent quality.
Focusing on MDR (Medical Device Regulation) compliance and ergonomic designs that meet high labor safety standards. Localized after-sales through regional partners.
Priority on durability in humid/tropical climates and ease of maintenance in remote diagnostic centers. Competitive pricing without compromising core safety.
High-end ICU platforms with smart connectivity and comprehensive digital integration for modern, high-traffic medical facilities.
Our global network spans across: Africa, Latin America, The Middle East, Southeast Asia, Europe, and North America. Through reliable products and professional services, we have earned the trust of international clients.

IQC Inspection

Processing

Marking

IQC

Processing
We are dedicated to making procurement simple, efficient, and worry-free. Our ecosystem includes:
Most reputable exporters like Medovia Medical provide ISO 13485 for medical device quality management and CE marking for the European market. For the US market, FDA registration is also standard.
We utilize high-duty cycle actuators that undergo rigorous testing (e.g., 10,000+ cycles under full load) to ensure they withstand the intensive environment of an ICU or ward.
Yes, we offer customization in terms of frame dimensions, siderail styles, mattress materials, and integrated scale systems for patient monitoring.
Depending on the quantity and customization, the average manufacturing lead time is 20-35 days, with efficient sea or air logistics arranged by our one-stop service team.
We provide detailed technical manuals, video training, and a supply of critical spare parts with every shipment. For large projects, we offer on-site technical support through local partners.