Medovia Medical
Medovia Medical
 Medovia Medical
Medovia Medical
The global respiratory care market is undergoing a paradigm shift. Driven by an aging population, rising prevalence of chronic obstructive pulmonary disease (COPD), and the post-pandemic emphasis on critical care infrastructure, the demand for high-precision respiratory devices has reached unprecedented levels. As a leading China Wholesale Respiratory Devices Factory, Jiangsu Medovia Medical Co., Ltd. sits at the intersection of technological innovation and manufacturing excellence.
The shift from reactive treatment to proactive respiratory management requires devices that are not only powerful but also portable and data-enabled. Our factory integrates IoT capabilities into anesthesia and ventilation systems to ensure real-time patient monitoring.
As a key Exporter, we mitigate global supply chain risks by maintaining vertically integrated manufacturing processes in Jiangsu, ensuring that hospitals in Europe, Africa, and Southeast Asia receive critical equipment without delay.
Navigating MDR (Medical Device Regulation) in Europe and FDA requirements in the Americas is central to our export strategy. We provide comprehensive documentation to support seamless localized registration.
Jiangsu Medovia Medical Co., Ltd. is a professional and reliable supplier in the medical equipment industry in China, committed to delivering advanced, high-quality, and cost-effective healthcare solutions to customers worldwide. With years of industry experience and continuous development, we have built a strong reputation as a trusted partner for medical institutions and distributors across the globe.
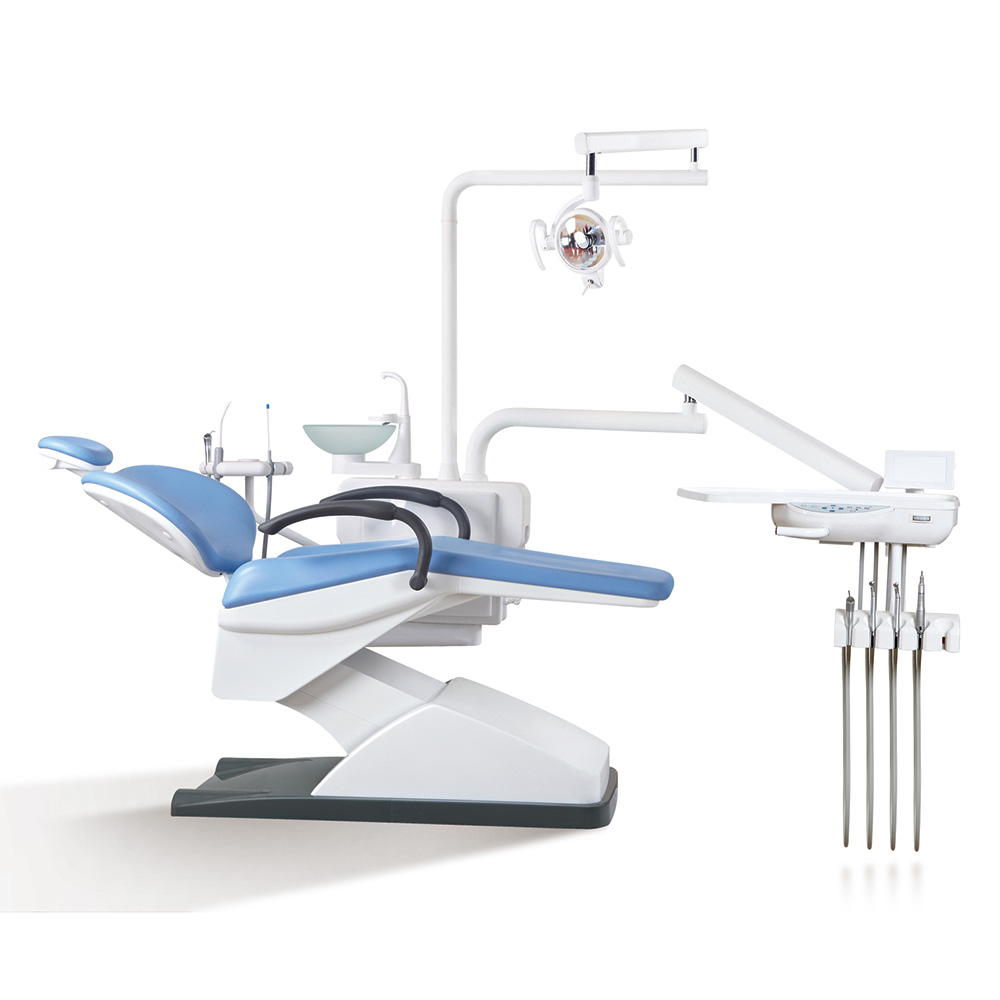
Our comprehensive product portfolio covers a wide range of medical applications, including surgical equipment, digital and portable X-ray systems, laboratory devices, patient monitors, ECG machines, ultrasound scanners, dental equipment, and hospital furniture.
Transitioning from bulky bedside units to ultra-portable, high-performance devices for emergency transport and home care. Example: Our Portable Ultrasound and Mini Centrifuges.
Implementing machine learning algorithms to predict respiratory failure and automate anesthesia dosage adjustments based on real-time biometric feedback.
A unified digital platform connecting ECG, Patient Monitors, and Respiratory devices to a centralized hospital Cloud for 5G-enabled remote diagnosis.
High-end anesthesia machines and multi-parameter monitors designed for stable performance in high-acuity environments.
Rugged, battery-operated AEDs and portable diagnostic tools for field hospitals and ambulance services.
Advanced blood cell counters and centrifuges for rapid respiratory infection screening and blood gas analysis.
Cost-effective ENT endoscopes and specialized furniture optimized for small-scale surgical procedures.
Quality is at the core of everything we do. Our factory utilizes CNC precision, laser engraving, and ultrasonic clearing to ensure every component of our medical devices meets the rigorous standards of the modern medical field.








We sincerely welcome customers from all over the world to visit our company and explore cooperation opportunities. Jiangsu Medovia Medical Co., Ltd. is committed to providing high-quality products, attentive service, and competitive prices, ensuring a truly win-win partnership.